
WORK PACKAGE 4 - Hydrogen Production
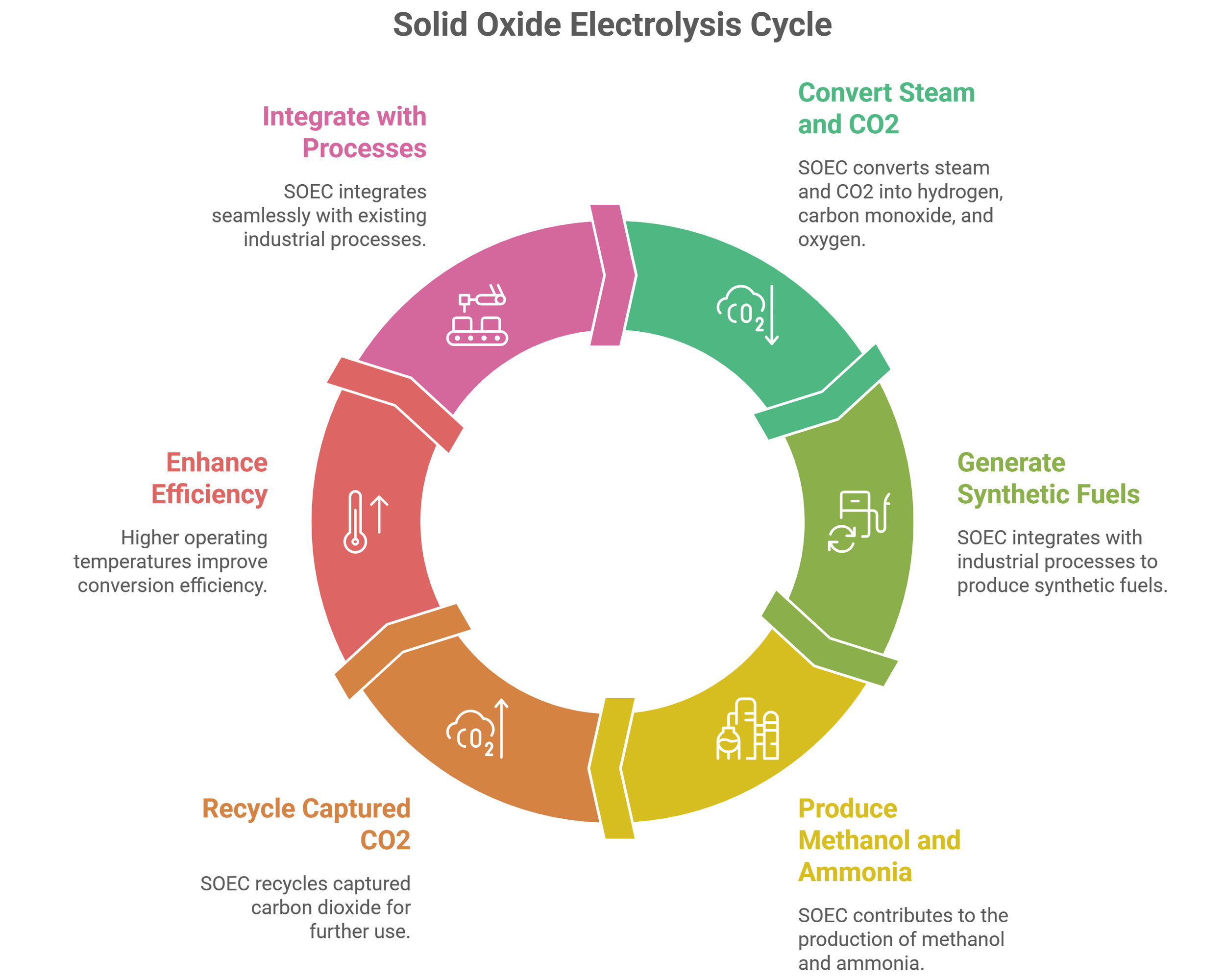
In addition this system can be used to produce syngas (electrolysis of steam and carbon di oxide). Their half-cell reaction involves, hydrogen evolution on steam side and oxygen evolution on the other side, with the electron movement through the electrode pathway and oxide ion movement along the ceramic membrane pathway.
A process diagram of single cell 10 x 10 cm2 - solid oxide electrolysis system, being developed in Laboratory of Chemical Technology campus, in Ghent University, Belgium is given above.


PROCESS DIAGRAM

